TM registration & COVID-19 vaccines
Let us start with an examination of the relationship between trademark registration and actual use of COVID-19 vaccines. In general terms, trademark registration is strongly recommended prior to the market launch of a new product / service as it provides effective protection against infringement and copycat attempts. This is especially true in the context of life saving COVID-19 vaccines as any potential infringement could undermine the public’s trust in the given vaccine or even lead to disastrous health outcomes.
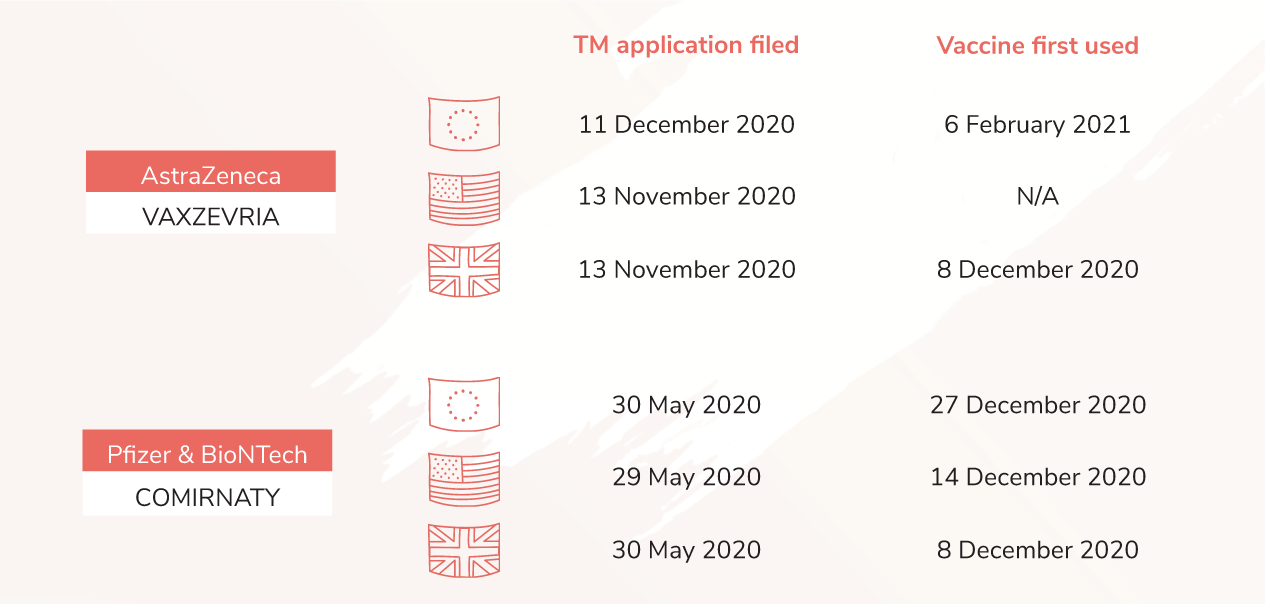
With the exception of Sputnik V, a clear pattern between trademark registration and the actual use emerges for all COVID-19 vaccines. An example can be made of Vaxzevria, an adenovirus vaccine developed by AstraZeneca. A trademark application in the UK was filed on 13th November 2020 and its actual use started from 4th January 2021. The trademark application in the EU was filed a month later (11th December 2020), with the actual use being also postponed by one month in the EU (6th February 2021). A similar effect can be found in the context of Comirnaty, an mRNA-based vaccine from Pfizer & BioNTech. Trademark applications in the EU, UK and US have been all filed in May 2020, and the date of the use of this vaccine in individual regions is also virtually the same (December 2020).
Sputnik V, a recombinant adenovirus vaccine developed by Gamaleya Research Institute, seems to be an exception to the aforementioned logic of registering a trademark prior to the medical clearance and consequent practical use of the vaccine. The trademark application in Russia was filed only 1 day prior to the medical approval of the vaccine, with the trademark including words “The First Registered COVID-19 Vaccine” signalling the significance of this achievement to the world. WIPO (World Intellectual Property Office) trademark application was filed one month later for Sputnik V, covering a range of countries including Brazil, Indonesia, India, Mexico, Oman, Philippines, Turkey, Armenia, Azerbaijan, Belarus, China, Cuba, Kyrgyzstan and the United States. With the exception of the United States, Sputnik V has been the primary COVID-19 vaccine used in the aforementioned countries.
This vaccine originating in Russia has attracted a considerable amount of attention over the recent weeks, particularly in the context of the European Union. Instead of seeking the medical clearance from the EU authorities, Sputnik V has been distributed to individual countries of the EU, starting with Hungary. This approach also coincides with the trademark strategy used for Sputnik V, where instead of a single application to the EUIPO (European Union Intellectual Property Office), a single application in the Hungarian jurisdiction has been filed.
COVID-19 & entrepreneurial minds

Over the last year, hundreds of COVID-19 related trademark applications have been filed with the intellectual property offices across the world. The common feature of these trademark attempts lies in the underlying entrepreneurial intentions, with individual applicants seeking to build on the wide awareness of the term and its associations. This is nothing new and a similar situation occurred in the aftermath of the tragic Charlie Hebdo shooting in Paris in 2015. Building on the strong recognition of “je suis Charlie” slogan, several trademark applications have been filed with the goal of monetising on this opportunity. In the context of COVID-19, trademark applications for the classes of clothing, personal items, educational materials and support groups have been the most popular. The vast majority of these trademark attempts can be however expected to be rejected by the respective intellectual property offices for failing to meet the criterion of distinctiveness.
What people really think about COVID-19?

Interestingly, trademark applications also provide an insight into the true feelings of the public about COVID-19. The use of obscured pejorative words is very common in these trademark attempts. While these applications seem to provide a valuable insight about what people really think about COVID-19, the success of these trademark attempts is expected to be very low, not only for failing to meet the criterion of distinctiveness but also for including pejorative words.
Summary & learning points
So what lessons can we take from COVID-19 trademark wars?
The best practice highlights the crucial importance of trademark registration prior to market launch of a new product / service
“Popularity” or strong associations of the public attract entrepreneurial minds that are very quick to exploit any emerging opportunity
It is always beneficial to rely on a trusted partner throughout the trademark registration process and carry out a thorough assessment of the trademark’s eligibility and similarity screening in order to avoid unnecessary costs


